Large pharmaceutical companies and scientific institutes, which began the development of vaccines against coronavirus earlier than anyone else, could become hostages of a pre-selected scheme, which may later turn out to be less effective, Ancha Baranova, professor at the School of Systems Biology at George Mason University (Virginia, USA), told Izvestia on July 30 …
On the eve of the Assistant Minister of Health of Russia, Alexei Kuznetsov, on July 29, announced that the coronavirus vaccine, which was developed at the Gamaleya Center for Epidemiology and Microbiology, is at the stage of state registration.
“At the very beginning, when SARS-CoV-2 was discovered, it became clear that on its surface there is an S-protein, which serves as a receptor. It sticks out of the virus. The very first idea that comes to mind in such a situation is to create antibodies to this S-protein and eliminate the virus, ”said Ancha Baranova.
The logic was that molecules of other types of coronavirus have other internal proteins that almost do not elicit an immune response. Therefore, scientists have made a logical conclusion that in the new coronavirus SARS-CoV-2, these internal proteins will also not cause an immune response. For this reason, many have relied on the S-protein, the biologist noted.
Including the Russian Center for Epidemiology and Microbiology named after Gamaleya began to make antibodies to the S-protein. And then there was evidence that the N-protein is a vulnerability of SARS-CoV-2. It, unlike the S-protein, is almost not susceptible to mutations, while being still available for antibodies.
Compared to N-protein, S-protein has disadvantages as an antigen for vaccination, she said. Indeed, mutations do occur in the S-protein. The vaccine will work worse against the mutant virus, because now antibodies, by definition, are loosely attached, Baranova added.
“I believe in the N-protein vaccine. My intellectual bet is on M-RNA vaccines because they induce stronger immunity. Secondly, based on N-protein. In Russia, I see the Biocada vaccine as such candidates. In America, the most advanced in this area are Pfizer and Moderna, ”the specialist noted.
At the same time, this does not close the possibilities for using the vaccine developed at the Gamaleya Center for Epidemiology and Microbiology, the scientist believes. This vaccine will most likely be made earlier, but in the future it will still be necessary to switch to more “durable” technologies, Ancha Baranova concluded.
Earlier, on July 27, it became known that the second phase of clinical trials of the Russian vaccine against coronavirus, developed by the Gamaleya Center, is at its final stage.
The number of confirmed cases of coronavirus infection in the world exceeded 17 million by July 30, according to Johns Hopkins University. In the top three in terms of cases: the United States (4.4 million), Brazil (2.5 million), India (1.5 million).
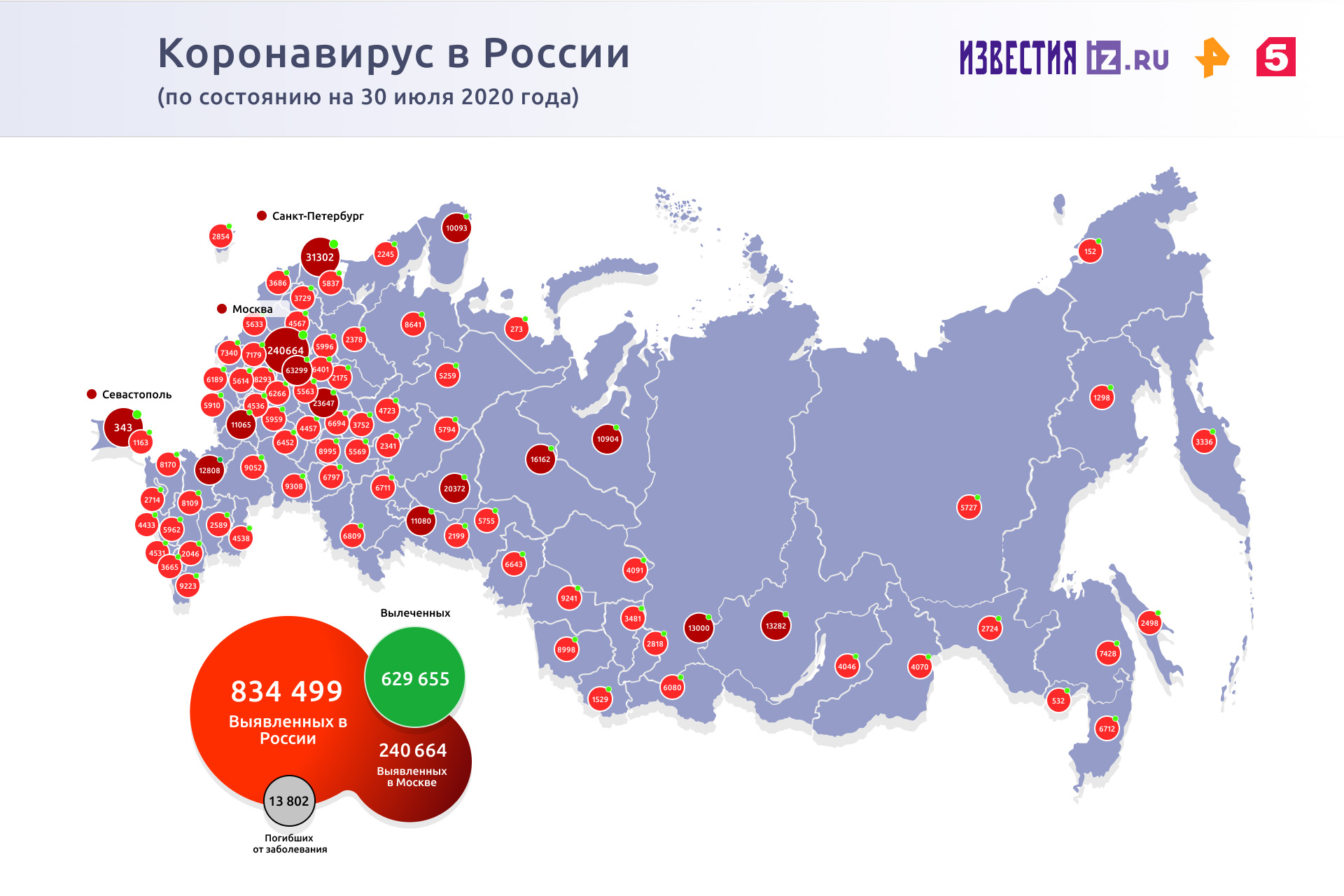
Russia is in fourth place. In the country, the total number of detected cases of COVID-19 has reached 834,499 people, 13,802 patients have died, and 629,655 recovered.
All information on the situation with the coronavirus is available on the websites of stopcoronavirus.rf and access vsem.rf. The hotline for the coronavirus is 8 (800) 2000-112. In addition, information is available under the hashtag #WeWeTogether.